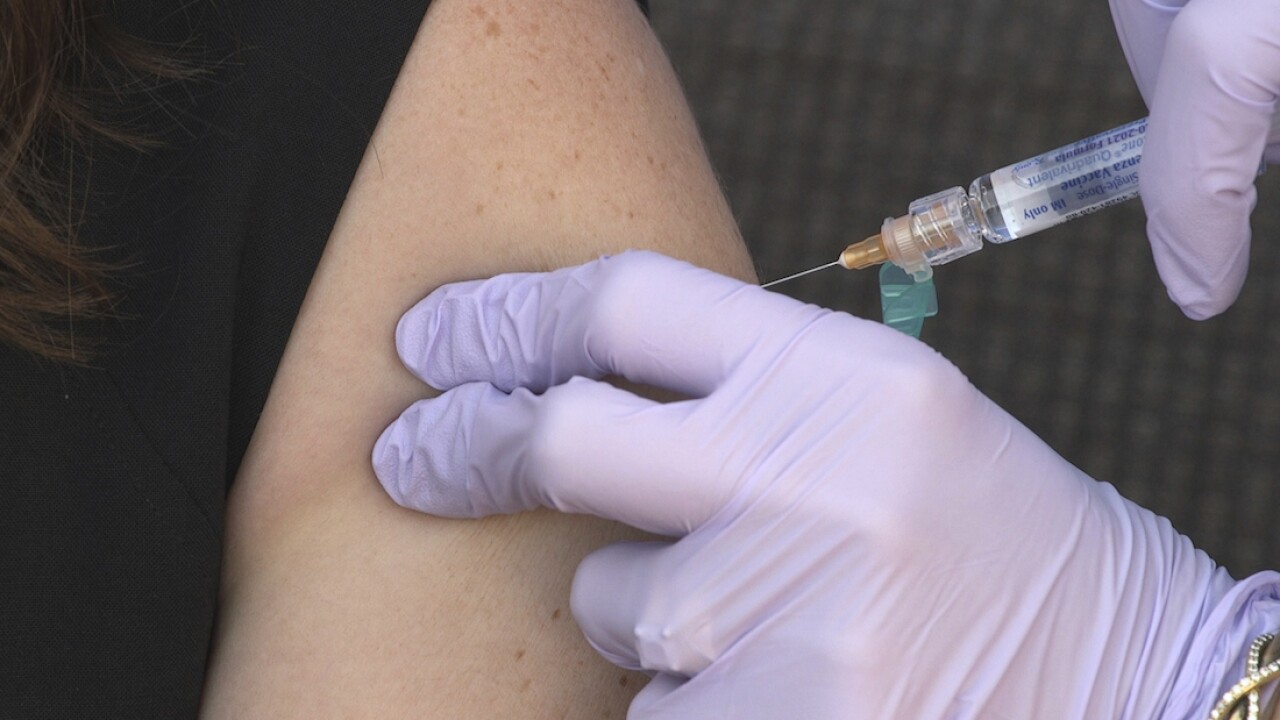
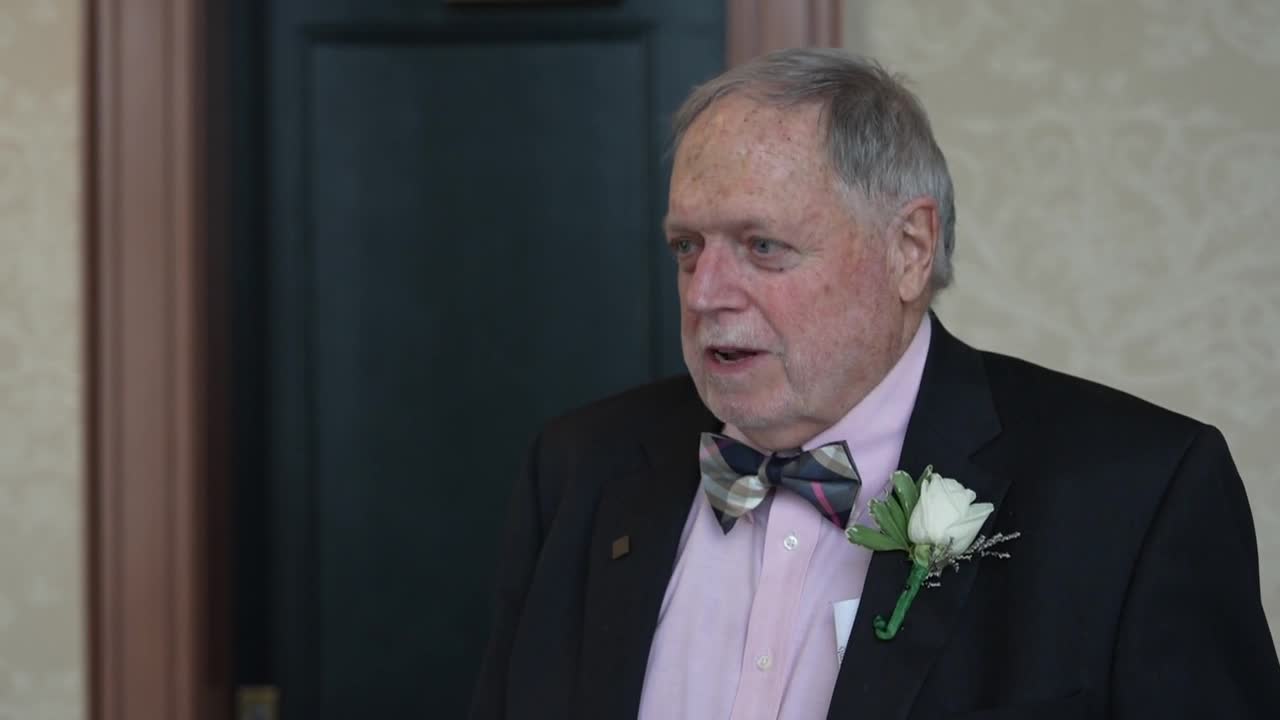RICHMOND, Va. -- The U.S. Food and Drug Administration is expected to authorize Pfizer's COVID-19 vaccine for children between the ages of 12 and 15 as early as next week, according to a federal official familiar with the authorization process.
This announcement comes one month after the company found that its vaccine, which is already authorized for teens over the age of 16, provides protection for younger adolescents.
Virginia Vaccine Czar, Dr. Danny Avula said 45% of the state's population has received at least one dose of the COVID-19 vaccine and efforts to vaccinate children will have a bigger impact in reducing virus cases in the commonwealth.
"That will open up about 400,000 additional Virginians who are eligible to be vaccinated," Avula says.
Pfizer studies have shown that children experience similar side effects to the vaccine as young adults, including pain, fever, chills and fatigue, particularly after the second dose.
While younger people are at a dramatically lower risk of serious side effects from COVID-19, they have made up a larger share of new virus cases across the U.S. as indoor activities and contact sports have resumed.
"As we look at outbreaks that are going on in Michigan and Minnesota, it's school-aged children, it's high school kids and college kids," Avula said. "That's had implications because in those states we're also seeing the highest rates of hospitalizations in the 30 to 40 age group that we've seen at any point in this pandemic."
But in the absence of long-term studies on vaccine safety and efficacy, some parents are skeptical about getting the vaccine for their children.
Avula says health department officials are reaching out to more pharmacies, primary care physicians and pediatricians, to help with mass distribution efforts involving children.
Recent surveys have shown several people have more trust in their own healthcare providers when making decisions about vaccinations, especially with their children.
"We've spent years building these relationships and learning about their children," said Richmond-based Pediatrician Dr. Mark Grabill.
Grabill says his group practice, Pediatric Associates of Richmond, is prepared to extend vaccinations to younger children under the age of 16 once FDA approval is granted.
However, like most physicians, he's waiting to see the full results of Pfizer's study and hear from the Federal Vaccine Advisory Committee, before discussing recommendations with his patients.
"Pediatricians are here to help parents and talk you through every stage of life," Grabill says. "This is no exception so please don't hesitate to call your pediatrician, ask for help, ask for advice and trust us, not the internet."
Avula says health officials plan to meet with school superintendents on May 11 to discuss the idea for schools to serve as possible vaccination sites. He says FDA approval could come in time for school-aged children to receive the vaccine in school before the end of the 2020-21 school year. While parents would not need to be present, parental authorization would be required.